
However, the model assumed electrons in the shells didn't interact with each other and couldn't explain why electrons seemed to stack in an irregular manner. It also explained why the noble gases were inert and why atoms on the left side of the periodic table attract electrons, while those on the right side lose them. For example, the shell model explained why atoms got smaller moving across a period (row) of the periodic table, even though they had more protons and electrons. The model explained some of the atomic properties of heavier atoms, which had never been reproduced before. Thus, the Bohr model for heavier atoms described electron shells. Once the level was full, additional electrons would be bumped up to the next level. 
Bohr proposed that electrons do not radiate energy as they orbit the nucleus, but exist in states of constant energy that he called stationary states. Bohr believed each electron orbit could only hold a set number of electrons. The Bohr Model has an atom consisting of a small, positively charged nucleus orbited by negatively charged electrons. Bohrs Atomic Model Following the discoveries of hydrogen emission spectra and the photoelectric effect, the Danish physicist Niels Bohr (1885-1962) proposed a new model of the atom in 1915. More electrons were required to cancel out the positive charge of all of these protons. He said the work has potential applications in next-generation computers and in the study of classical and quantum chaos.Heavier atoms contain more protons in the nucleus than the hydrogen atom. Draw an arrow that indicates the dot has moved from the calcium to the chloride. (b) The energy of the orbit becomes increasingly less negative with increasing n. 2: The Bohr Model of the Hydrogen Atom (a) The distance of the orbit from the nucleus increases with increasing n. Draw another dot on the outside ring of the chlorine atom on the left. In this state the radius of the orbit is also infinite. 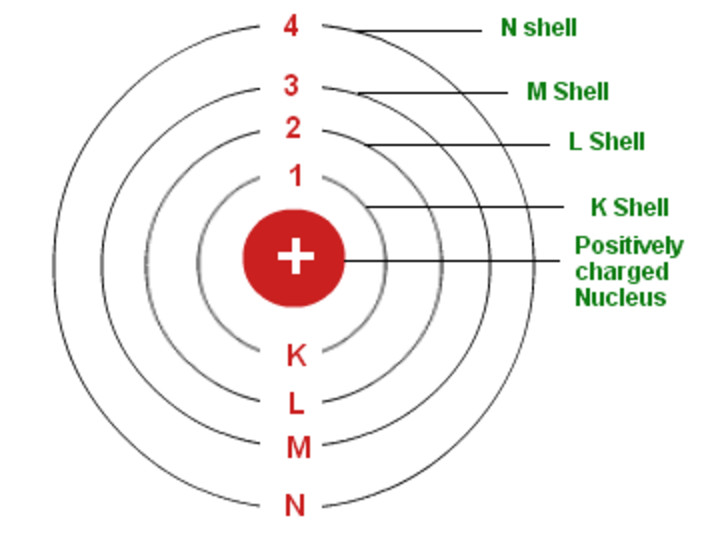
One of the fundamental laws of physics is that matter is most stable with the lowest possible energy. The lowest few energy levels are shown in Figure 3.2.1 3.2. The more negative the calculated value, the lower the energy. "Our measurements show that the electrons remain localized for several orbits and behave much as classical particles," Dunning said. Remove one dot from the bottom calcium atom’s outside ring at the 9 oclock position. 1: Quantum numbers and energy levels in a hydrogen atom. In fact, the atoms are true atomic giants, with diameters approaching one millimeter. Using a carefully tailored series of short electric pulses, the team was then able to coax the atoms into a precise configuration with one point-like, "localized" electron orbiting far from the nucleus. Using lasers, the researchers excited potassium atoms to extremely high levels. The team included members from Oak Ridge National Laboratory and Vienna University of Technology. The protons and neutrons are placed inside the nucleus while the electrons are housed in three shells i.e. "Using highly excited Rydberg atoms and a series of pulsed electric fields, we were able to manipulate the electron motion and create circular, planet-like states." Conclusion The final Bohr model of the Sulfur atom consists of 16 protons, 16 neutrons, and 16 electrons. "In a sufficiently large system, the quantum effects at the atomic scale can transition into the classical mechanics found in Bohr's model," said lead researcher Barry Dunning, Rice's Sam and Helen Worden Professor of Physics and Astronomy. But his notion of electrons traveling in discrete orbits was eventually displaced by quantum mechanics, which revealed that electrons don't have precise positions but are instead distributed in wave-like patterns. Bohr's model led to a deeper understanding of both the chemical and optical properties of atoms and won him a Nobel Prize in 1922. The research is available online in Physical Review Letters.īohr offered the first successful theoretical model of the atom in 1913, suggesting that electrons traveled in orbits around the atom's nucleus like planets orbiting a star. HOUSTON - J- Nearly a century after Danish physicist Niels Bohr offered his planet-like model of the hydrogen atom, a Rice University-led team of physicists has created giant, millimeter-sized atoms that resemble it more closely than any other experimental realization yet achieved. Image: Using laser beams and electric fields, Rice physicists coaxed a point-like, "localized" electron to orbit far from the nucleus of a potassium atom.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
